The biology behind NK cell immunotherapy
NK cells sit at the intersection of innate immunity and translational oncology. We approach their biology with scientific discipline and regulatory awareness.
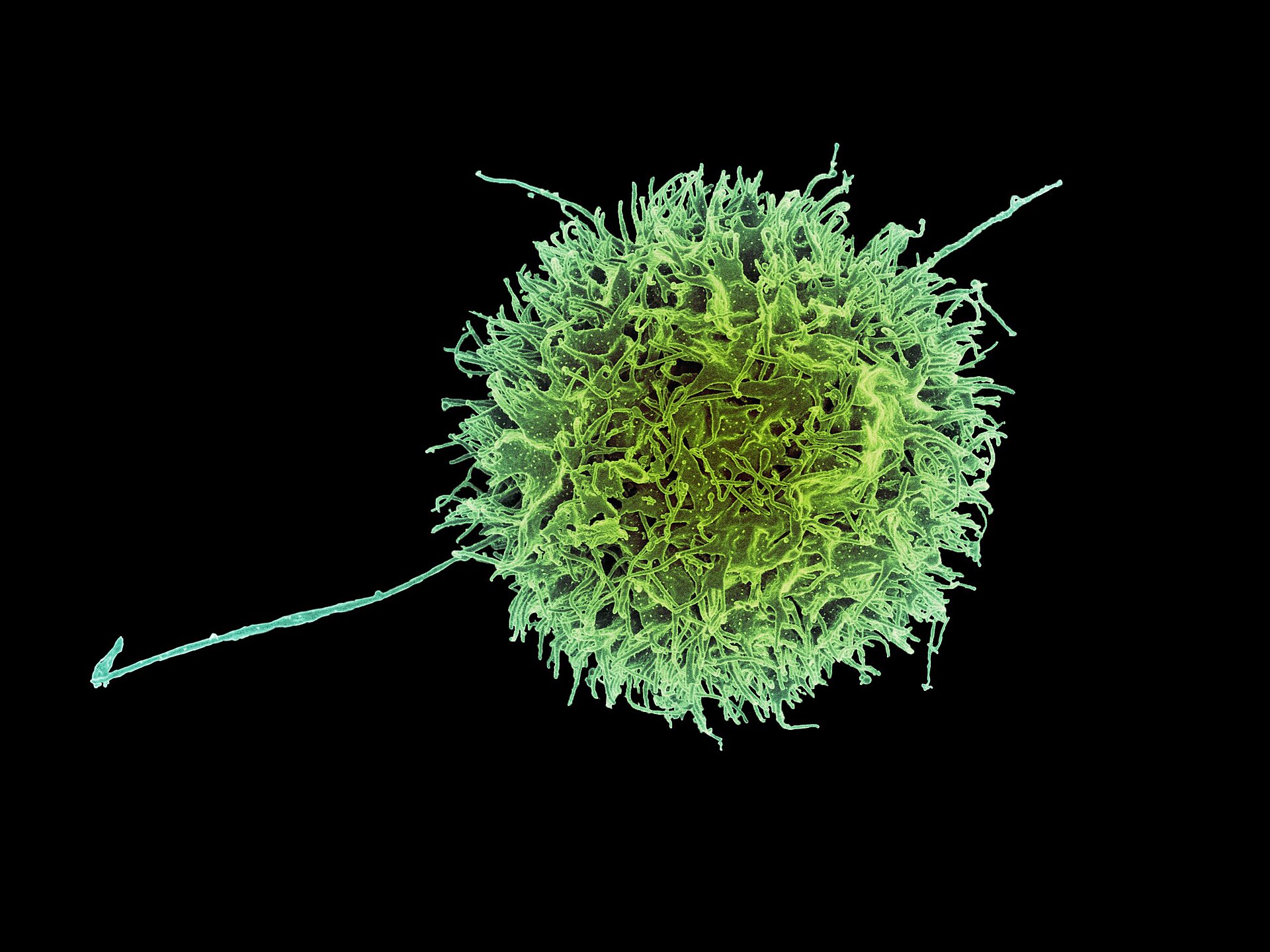
A single cell at the center of innate tumor recognition
Natural killer cells operate without prior antigen sensitization, integrating activating and inhibitory signals to discriminate stressed or transformed cells from healthy tissue. Their morphology — granular cytoplasm, dynamic surface receptors, and rapid cytolytic machinery — underpins the therapeutic rationale we develop at BioNK.
Innate immune effector cells
Natural killer (NK) cells are innate immune effectors equipped with cytotoxic granules, cytokine signaling capacity, and a balanced repertoire of activating and inhibitory receptors. Their ability to recognize stressed or transformed cells without prior antigen sensitization positions them as a foundational modality in advanced immunotherapy research.
Why NK cells matter in oncology
NK biology offers innate tumor recognition, potential allogeneic use, antibody-dependent cellular cytotoxicity, and compatibility with engineering strategies. Where scientifically supported, NK cells may be combined with antibodies, checkpoint strategies, cytokines, or other immune modulators to extend therapeutic reach.
Acknowledging the development complexity
Tumor microenvironment suppression, trafficking and infiltration into solid tumors, persistence, antigen heterogeneity, potency assay complexity, manufacturing consistency, cryopreservation sensitivity, and on-target/off-tumor risk for engineered products all require disciplined translational planning.
Translational rigor over hype
BioNK prioritizes translational assays, transparent potency rationale, model limitations, regulatory-grade documentation, and clinical validation. Evidence generation is ongoing and clinical development remains subject to applicable regulatory and ethics approvals.
Information on this page is provided for scientific and educational purposes. It does not constitute medical advice or claims of efficacy. BioNK programs are investigational and remain subject to regulatory, ethics, CMC, nonclinical and clinical validation.